There is this old, ever repeating view of humanity as a counter-productive force when it comes to nature conservation, and that action toward a sustainable world is therefore doomed to failure right from the start. This view seems tempting, since the reasoning makes sense at first sight. Even if we increased material efficiency noticeably, so it goes, the drastic growth in material demand will override any achievements. This view is what Julian Allwood and Jonathan Cullen called “with one eye open”. They wrote a comprehensive book about sustainable materials called “With Both Eyes Open,” available online. And that’s the interesting part. “Both Eyes Open” explains that the assumptions the “One Eye” viewpoint is based on are insufficient. This article summarizes the main findings of the book, namely how civilization can reduce its material need without compromising the comfort level we’ve gotten used to (and citizens of emerging economies strive to get used to, too) using the example of global aluminum and steel consumption. Once you adjust your assumptions to these new ways of thinking, you’ll find that all are proven wrong who think cutting emissions by 50% by 2050 is impossible, given that in the same period the demand for raw materials is to double.
Steel Demand Constant in Europe and US, but Doubling Globally
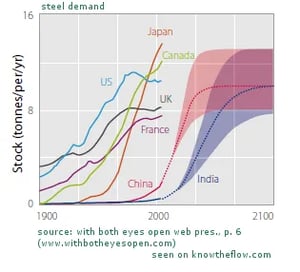 Starting roughly in the mid-1970s, most western industrial nations saw their demand for steel stabilize. Even though the economy grew constantly a stable level of raw material consumption was reached at a certain point. Assuming China and India, the biggest growing economies and developing countries in the world, catch up with their demand, by 2050 they will have reached a similar per-capita level as the western nations. As the following graphic shows, the Indian and Chinese steel demand will hover around the European and North American levels. France, for instance, uses 8 tons per capita annually, whereas Japan never stopped increasing its demand, reaching 14 annual tons per capita today.
Starting roughly in the mid-1970s, most western industrial nations saw their demand for steel stabilize. Even though the economy grew constantly a stable level of raw material consumption was reached at a certain point. Assuming China and India, the biggest growing economies and developing countries in the world, catch up with their demand, by 2050 they will have reached a similar per-capita level as the western nations. As the following graphic shows, the Indian and Chinese steel demand will hover around the European and North American levels. France, for instance, uses 8 tons per capita annually, whereas Japan never stopped increasing its demand, reaching 14 annual tons per capita today.
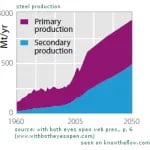 As a result, global steel production will have to double in the next 40 years. It is out of question, however, that doubling production could be allowed to double the related emissions at the same time. We already emit more than the atmosphere can take. Rather than increasing, we should be cutting our emissions by 50%, as recommended by the IPCC. So what can we do? Yes, recycling is a good idea. The following graphic shows how to match increased demand while keeping the production constant – recycling, i.e., secondary production, is the key.
As a result, global steel production will have to double in the next 40 years. It is out of question, however, that doubling production could be allowed to double the related emissions at the same time. We already emit more than the atmosphere can take. Rather than increasing, we should be cutting our emissions by 50%, as recommended by the IPCC. So what can we do? Yes, recycling is a good idea. The following graphic shows how to match increased demand while keeping the production constant – recycling, i.e., secondary production, is the key.
Where is it Consumed? Steel Flows From Raw Material to Product
 This Sankey diagram (on the right) tells you everything. It shows the mass flows of steel, from the raw iron ore to the final car, and every step of material conversion in between. Basically, there are two types of inputs to make steel. One, of course, is the iron ore, the true raw material. The other input is scrap. Scrap, in part, consists of leftovers from the production process. In the diagram it is marked as forming and fabrication scrap. Then, there is end-of-life scrap. This is the recycling part. End-of-life – as in post-consumer, things we dispose. The latter makes up less than half of the scrap input and could be increased a lot. So we learn that the potential for steel recycling has not yet been much exploited.
This Sankey diagram (on the right) tells you everything. It shows the mass flows of steel, from the raw iron ore to the final car, and every step of material conversion in between. Basically, there are two types of inputs to make steel. One, of course, is the iron ore, the true raw material. The other input is scrap. Scrap, in part, consists of leftovers from the production process. In the diagram it is marked as forming and fabrication scrap. Then, there is end-of-life scrap. This is the recycling part. End-of-life – as in post-consumer, things we dispose. The latter makes up less than half of the scrap input and could be increased a lot. So we learn that the potential for steel recycling has not yet been much exploited.
Another fact that can be derived from the diagram is that one fourth of the liquid steel is scrapped during its production. Improved process and material flow management could save a lot of material here. We’ll come back to that later.
Energy Efficiency Potential in Metal Production Almost Reached
First, since energy contributes around 35% of the cost of making both steel and aluminum, note that serious energy efficiency measures have already been taken in recent years. Back in the 1970s, in order to produce one ton of crude steel you needed more than 50 GJ of energy. The modern average is half that amount, as you can see in the following graphic. Of course, there is still room for improvement, as best practice shows. The ideal way to produce one ton of crude steel consumes 50% less energy than the average method. Not bad, but the goal of reducing overall emissions by 50% by 2050 cannot be reached even if best practice spreads. Rather, we would need to reduce emissions for steel production by 75%! Why? That’s easy – although cutting emissions by 50% would be great, since demand for steel will double, we don’t need a half, but rather a quarter of the previous emissions.
Of course, there is hope for a rising share of renewables. And numerous studies have shown that 100% renewable energy generation by 2050 is not only feasible, but also beneficial for the economy. However, conventional energy lobbies are strong and we shouldn’t forget that one kWh saved is immeasurably better than one newly generated, be it renewable or not. And this truth is vital. You can directly apply it to material efficiency, too. One ton of material saved is much better than one substituted with a renewable material.
Improve Design, Use Less Material
Now, what are the strategies to reduce material consumption? How do we match the rising global demand and achieve a 50% emission reduction? Allwood and Cullen (remember? The authors of the “Both Eyes Open” book?) proposed a mix of ideas. Some are surprisingly obvious, some reflect all their expertise. The first idea is pretty well-known. What they called “use less material by design” is commonly known as eco or lightweight design. From page 163 on, fabrication engineers will find detailed recommendations on how processing steel and aluminum products can be made more material efficient. Even though it goes beyond common knowledge, let me briefly quote five principles that save material in production:
- Support multiple loads together
- Don’t over-specify the loads
- Align loads with members to avoid bending
- Optimise the cross-section for bending
- Choose the best material
Allwood and Cullen call for a rewriting of standards to allow more appropriate construction components. Just to mention a couple of examples: instead of the standard I-beam, structural engineers should consider using open-web joists or cellular beams. These use less material by far but still meet the required capacity. However, sometimes the standards still call for I-beams.
But it’s not only about these details. Material efficient building design starts as early as the first sketch. To illustrate this point, the authors compared two similar-sized buildings developed for the London Olympics (see page 170). They came to the conclusion that the first building, the velodrome, followed an approach to reduce the embodied emissions of the building right from the start. This is how the demand for steel was reduced by almost one third, compared to the second building, the Aquatics Centre. Material optimization there was less of a concern than an iconic design.
The architects for the Velodrome had a vision to build a minimum structure building ‘shrink- wrapped’ around the sport and spectators. As a result the geometry was governed by the track layout and required sightlines; this ‘saddle’ shape allowed use of a lightweight cable-net roof system where the steel is used in tension to span 130 metres between supports. Despite initial concerns about costs and risks, the contractor could save money and time by using this system and the client approved. The cable-net roof saved 27 % of the steel that would have been required in an alternative steel arch option. An advanced dynamic analysis of the seating structure showed that combining the roof, stand and façade support systems, gave performance within accepted limits despite being lighter than code recommendations.
Improve Manufacturing Processes, Reduce Yield Losses
More than a quarter of the liquid metal used in manufacturing is scrapped. Why? Because blanking, trimming and machining produce leftovers. And because of facts that you’re only likely to know if you’re an industry insider or a good materials engineering student. Like the following one, explained on page 197:
When liquids solidify, they do so from outer surfaces towards their interior, and for liquid metal with complex compositions, during this process, the composition of the remaining liquid changes. As described in chapter 8, different cooling rates at the surface and centre of an aluminium ingot lead to a different, lower quality, microstructure and composition at the surface. As a result, 150 mm is currently sawn from the head and tail of each cast aluminium ingot and the outer 20 mm from the top and bottom surfaces is removed by ‘scalping’. (Scalping, which is also applied to the hair of new army recruits, is a large scale machining process.)
And that’s one of the reasons why Allwood and Cullen called for new forming and casting processes.
Re-Use, Don’t Re-cycle, Manufacturing Scrap
 What’s so bad about recycling? Well, recycling production leftovers by remelting the metal consumes energy and causes emissions, whereas re-using them is the most environmentally friendly option. There are two concrete examples for the use of manufacturing scrap. One is pretty simple – when blanking press skeletons haven’t been fully used, why not stamp smaller pieces? Allwood and Cullen showed that it’s possible to “exploit every last square millimetre of each sheet”, so the blanking skeletons are worthy of their name (see illustration on the right). The other example is aluminum swarf, used through solid bonding (see p. 204):
What’s so bad about recycling? Well, recycling production leftovers by remelting the metal consumes energy and causes emissions, whereas re-using them is the most environmentally friendly option. There are two concrete examples for the use of manufacturing scrap. One is pretty simple – when blanking press skeletons haven’t been fully used, why not stamp smaller pieces? Allwood and Cullen showed that it’s possible to “exploit every last square millimetre of each sheet”, so the blanking skeletons are worthy of their name (see illustration on the right). The other example is aluminum swarf, used through solid bonding (see p. 204):
Aluminium is a very reactive metal, and as we learned in chapter 3, under normal conditions ‘naked’ aluminium will rapidly and within milliseconds react (join) with oxygen atoms in the air to form a thin protective layer of aluminium oxide. However, if we were able to bring together two ‘naked’ surfaces of aluminium, with no oxygen present, the two surfaces would instead react with each other, and bond. So, pure aluminium will weld to itself at room temperature, and this gives us a chance to re-use aluminium swarf without melting.
Re-Use Metal Products Without Melting
The activity of taking an object others have wasted and giving it a new life is very common in favelas, slums and “poorer“ neighborhoods around the globe. As soon as it was dubbed upcycling a few years ago, more and more conscientious people, who could also afford buying new stuff, also found ways to take old consumer goods and transform them into unique art objects. Communities like we upcycle collect and display the most creative ideas.
However positive these actions are, it is by far not enough to recover big amounts of metal. Allwood and Cullen point out another beam example. Generally, before we demolish a building, in 99 out of 100 cases, the beams are still intact. Who says we cannot reuse them in another building? On page 219, the authors provide us with seven examples from Canada and the UK, where construction steel has been taken from a nearby demolition site and used in a new building, saving 0.8 – 1.8 kg CO2 per kg steel. Another idea is to change the design of train rails. At the end of their lifespan, their surface is finished, whereas the rest of the rail remains theoretically usable. For this reason, Allwood and Cullen propose a double-sided rail design. Embedded in concrete, they could get a second life when turned over and would last two times longer with the same consumption of material! The double-use rail design requires more concrete, but still performs better in terms of overall emission savings.
Products That Last Longer – Keep the Old Car or Not?
There is hardly a single environmentally conscious man in the western world who hasn’t justified driving his old car by mentioning the embodied emissions of a new, albeit more fuel efficient replacement. And guess what: Allwood and Cullen also have an innovative solution for this issue. Yes, getting rid of any car and commuting by bike or public transport would be the best thing, but that’s not what they talked about. Their idea, much rather, is to replace just the most emission-sensitive part of the car, i.e., the engine, instead of the whole car. They assumed that a newly produced mid-size car has embodied emissions of 6.3 t CO2. The annual emissions, at 20,000 km, depend on the engine. If you take a 5-year old car, with emissions of 128 g CO2/km, they add up to 2.56 t CO2 per year. A younger car emitting 90 g CO2/km saves you 0.76 t per year, but produces another 6.3 t during its production. The motor upgrade, however, only produces 0.9 tons CO2. As you see, upgrading the motor to a more efficient one, while keeping the rest of the car, saves both material and emissions and is without a doubt the best solution. This idea can be applied to many other uses. The more embodied the emissions (e.g. in a ship), the better it will work.
Reduce Final Demand Without Impoverishment
In order to convince you to read this marvelous eBook, and to make room for one of the most striking contradictions of our time, I allow myself to quote a paragraph from page 250. It is also the concluding paragraph for my article. Not because there’s nothing more to discover in the book, but rather because you should read it yourself instead of just scanning my few lines in the blog…
In public, no business leader and no politician or policy maker can propose an ambition of reduced profit or induced recession. But in private, after a glass of wine and a nice meal, virtually everyone we’ve talked to in those positions has said to us “of course, we all know we’re simply consuming too much.” And in developed economies we do all know that—because nearly all of us can remember consuming less a few years ago. If we read the gravestones in our local churchyard, we have yet to find one which says “Here lies John Smith, whom we remember because he owned a large pile of material”.




